Practical, hands-on support to ensure your trials runs smoothly at the site level.

About us
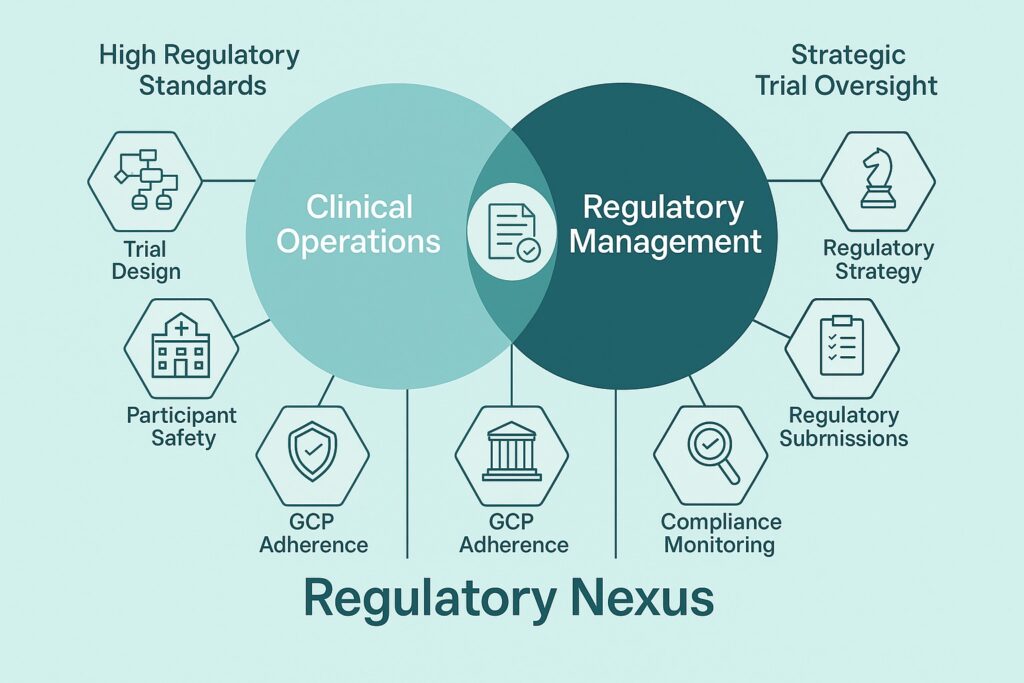
At Regulatory Nexus, we specialize in bridging the gap between sponsors and clinical trial sites bringing research opportunities directly to capable sites and helping sponsors find the right partners to execute their studies with precision and compliance. With expertise in Regulatory Affairs and Clinical Research Operations, we deliver services that shorten timelines, improve trial quality, and keep your study inspection-ready
SERVICES OFFERED
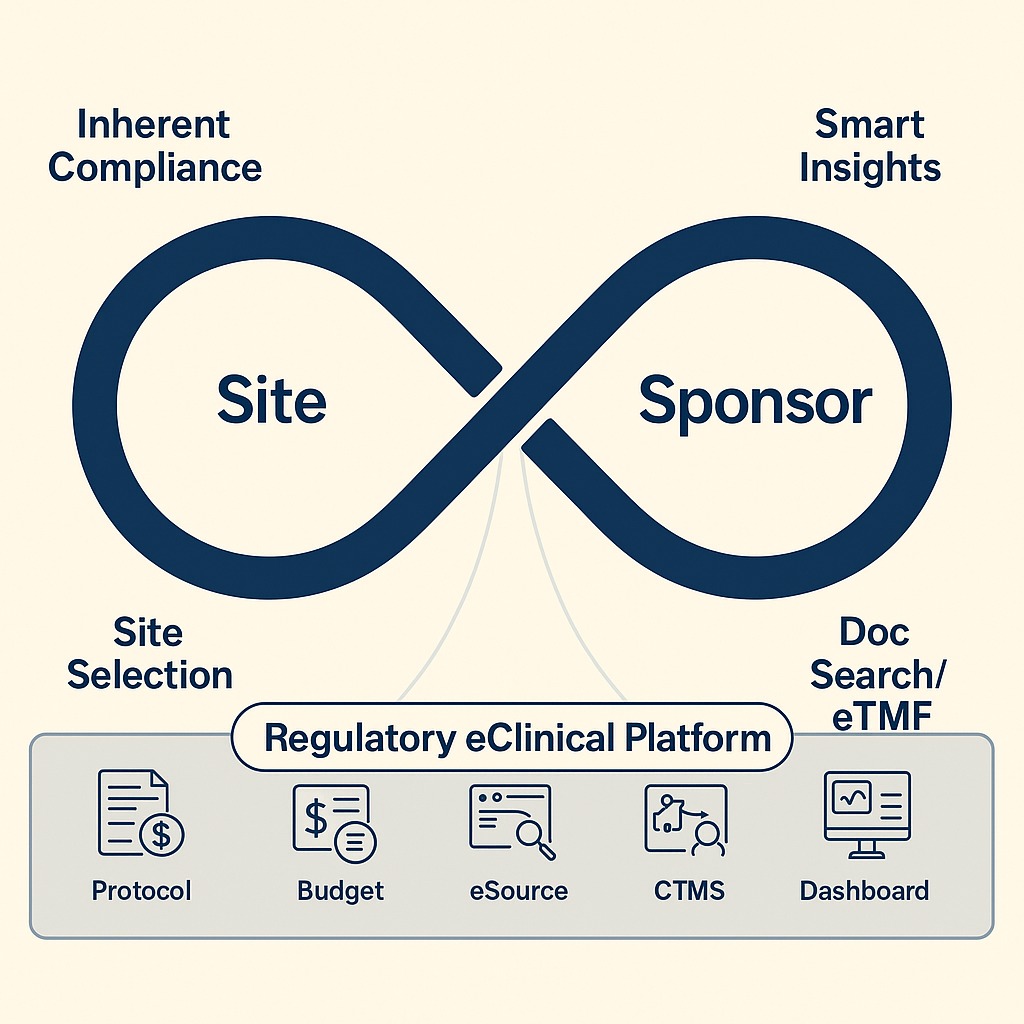
Sponsor–Site Partnership Solutions
We connect the right sponsors with the right clinical trial sites to create strong, results-driven partnerships
- Identifying and introducing sites with the right patient population and resources.
- Conducting site feasibility assessments to ensure readiness and recruitment potential
- Guiding sites through onboarding, document collection, and system setup.
- Acting as a dedicated liaison to ensure smooth communication and timely deliverables
Regulatory Affairs Support
Stay compliant with changing regulations and avoid costly delays
- Preparation and submission of clinical trial applications to ethics committees and regulatory authorities
- Expert review of protocols, informed consent forms, and Investigator Brochures for regulatory compliance
- Post-approval management of amendments, progress reports, and compliance updates.
- Continuous monitoring of regulatory changes to keep your trials on track
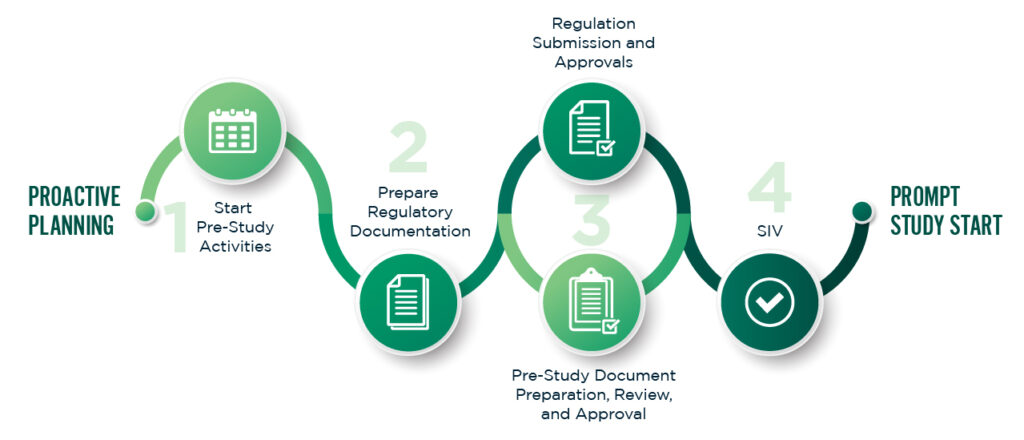
Clinical Operations Assistance
Practical, hands-on support to ensure your trial runs smoothly at the site level.
- Study start-up coordination, including regulatory green light processes
- Site staff training on GCP, protocol requirements, and trial systems
- Site performance optimization to boost patient recruitment and retention
About Founder

Rajashekhar Ittedi
I help clinical research sites operate with confidence, compliance, and efficiency. With a PharmD, a Master’s in Regulatory Affairs, and 3+ years leading site-level operations, I specialize in transforming complex regulatory and operational requirements into smooth, streamlined workflows. From feasibility and start-up to data accuracy, monitoring visit readiness, and FDA/GCP audit preparation, I ensure sites are fully equipped to deliver high-quality research with reliability and speed. My mission is simple: empower sites to perform at their best and elevate the standard of clinical trial execution
Documentation & Data Management Support
Secure, compliant, and efficient documentation services that reduce site burden and keep your trial inspection-ready.
Data Management
- Entry of CRF/eCRF data into sponsor-designated EDC systems (Medidata, Oracle InForm, Veeva, REDCap, Green Light Guru etc.) through secure portals.
- Timely, accurate transcription of data from completed source documents or paper CRFs.
- Timely, accurate transcription of data from completed source documents or paper CRFs.
Audit Readiness
- Performing double data verification to minimize discrepancies.
- Generating, tracking, and resolving queries in coordination with sites and sponsors.
- Continuous data cleaning to maintain a high-quality database ready for interim and final analyses.

Get in touch
Our Office
1215 E Vista Del Cerro Dr, Tempe, Arizona, 85281
info@regulatorynexus.in
Phone Number
+1 857 746 9255
